SureSelect CD HRR17
SureSelect HRR 靶向捕獲基因套組
Agilent is collaborating with SeqOne Genomics to deliver an analysis platform for the Agilent SureSelectCD HRR17 panel, enabling an accurate, streamlined, fully validated homologous recombination deficiency(HRD) workflow solution.
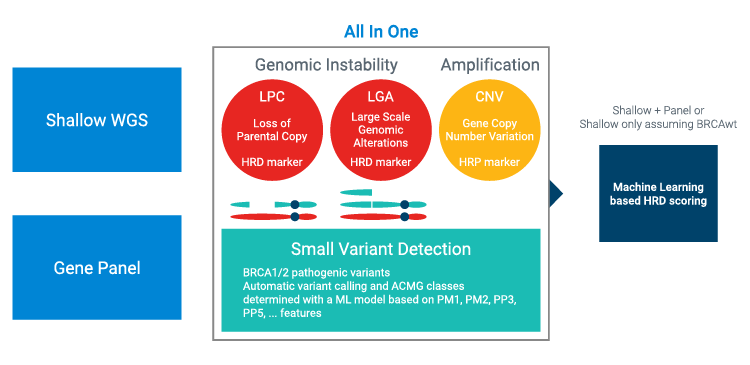
HRD is a key biomarker to identify ovarian cancer patients likely to respond to PARP inhibitor (PARPi) therapy. It can now be offered by any genetic lab routinely performing targeted capture, enabling labs to expand testing portfolios and bring tests closer to patients for improved sample efficiency and turnaround times. The SureSelect HRR17 gene panel has been developed to cover 17 genes associated with HRR or other genes of interest. Importantly, this panel can be combined with low-pass whole-genome sequencing (LP-WGS) and the analysis solution from SeqOne to provide a fully validated HRD workflow.


- Features a 17-gene panel, total footprint 114.3 Kb, specifically selected to identify BRCA and HRRg ene mutations and used with SeqOneHRD analysis
- Suitable with SureSelect XT HS or XT HS2 library preparation, including automation options for high throughput and a fully walkaway capture workflow
- Compatible with Illumina chemistry for32 libraries per NextSeq run (two x 150bp mid-output) or ten libraries per run for HRR panel and LP-WGS, delivering a complete SeqOne HRD solution
產品列表
| 貨號 | 產品 | 數量 | |
|---|---|---|---|
| 5282-0086 | SureSelect CD HRR17 16 | 16 rxns | |
| 5282-0087 | SureSelect CD HRR17 96 | 96 rxns | |
| 5282-0088 | SureSelect CD HRR17 96 Auto | 96 rxns Auto | |
| G9723A | Agilent Magnis SureSelect CD HRR17, ILM, 32 | 32 rxns | |
| G9723B | Agilent Magnis SureSelect CD HRR17, ILM, 96 | 96 rxns |
Building on the new Agilent CD HRR17 gene panel, SeqOne Genomics has developed a clinically validated HRD solution, designed for efficient development in labs that want to deliver HRD tests in-house. The workflow combines a targeted capture panel and LP-WGS that is flexible enough to accommodate a wide range of patient volumes and lab configurations. The solution delivers excellent performance, even with samples of low cellularity, and reduces the rate of inconclusive results.
回到頂端


